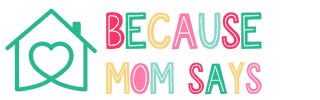Scientists have warned against relying too much on TikTok’s most recent “miracle” of weight loss after a troubling new study connected it to several infrequent but possibly lethal mental crises.
Ozempic’s Sought-After Side Effect of Weight Loss
Editorial credit: KK Stock / Shutterstock.
A prescription injection called Ozempic was created to help people with Type 2 diabetes control their blood sugar levels. Nonetheless, one desired side effect of the diabetic medication has contributed to its increased popularity: weight reduction. Ozempic is based on glucagon-like peptide 1 (GLP-1), a hormone found naturally in humans and crucial for controlling blood sugar and hunger. Semaglutide, a chemical that resembles the structure of this GLP-1 hormone and stimulates its receptors, is its active component.
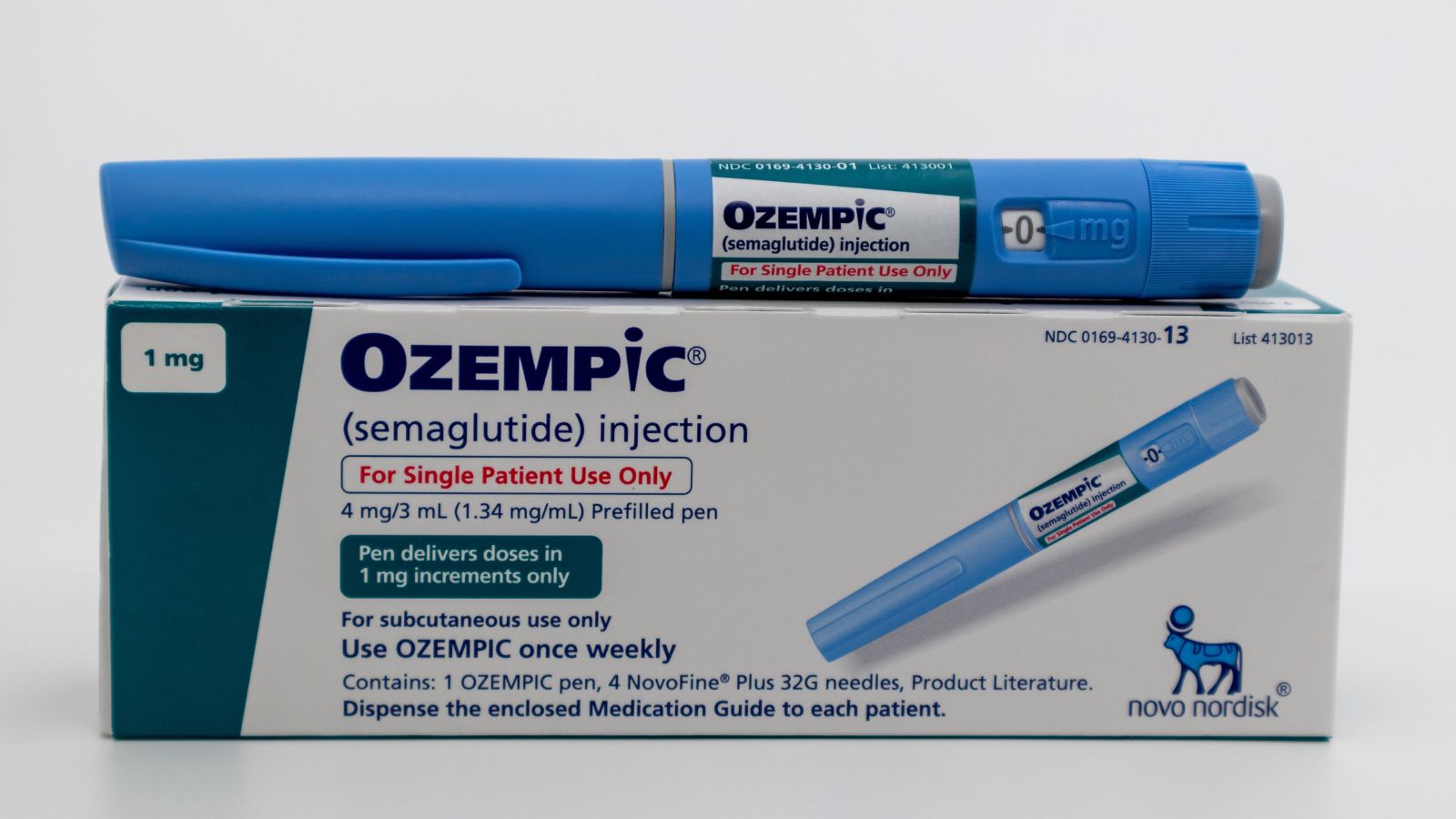
Ozempic’s Sister Drug, Wegovy
Editorial credit: KK Stock / Shutterstock.
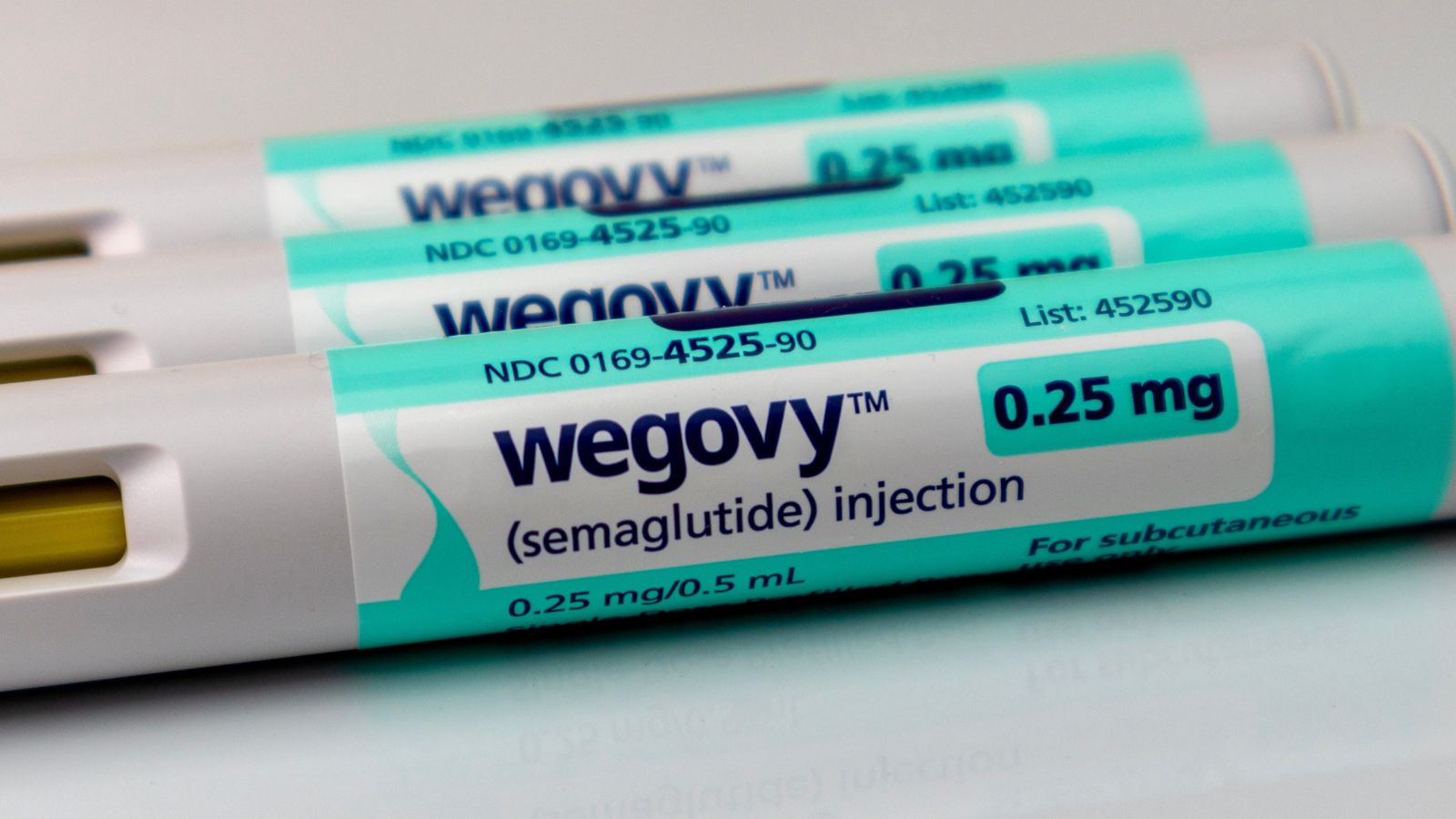
By turning on these receptors, semaglutide makes us feel fuller longer before our stomach empties, which reduces our hunger and decreases our propensity to overeat. Another medication that contains semaglutide is Wegovy, the sister product of Ozempic, an FDA-approved medication for long-term weight control. According to healthcare analytics company Trilliant Health, prescriptions for GLP-1 mimic drugs like semaglutide rose 300 percent nationwide between the beginning of 2020 and the end of 2022.
Many Uncomfortable Side Effects

Editorial credit: XanderSt / Shutterstock.
Celebrities and influencers have embraced the medication; on TikTok, #ozempic has over 1.4 billion views. However, semaglutide has been demonstrated to have some unpleasant side effects when taken as a medication for weight reduction.
Adverse Psychiatric Side Effects

Editorial credit: Michael Vi / Shutterstock.
The majority of research on the adverse effects of semaglutide has been on gastrointestinal issues; however, a recent study that was published in the International Journal of Clinical Pharmacy has revealed a worrying correlation between the medication and negative mental events, such as suicidal thoughts, anxiety, and sadness. The FDA was mentioned by pharmaceutical company Novo Nordisk, which makes Ozempic and Wegovy, when they said that no proof of suicidal thoughts or acts was discovered concerning these medications.
New Anti-Obesity Medications

Editorial credit: fizkes / Shutterstock.
Mansour Tobaiqy, an associate professor of clinical pharmacology at the University of Jeddah in Saudi Arabia, was the study’s first author. “We believe that our findings, which highlight potential mental health issues associated with new anti-obesity medications, are of significant importance to both healthcare providers and patients,” he said.
Assess a Patient’s Mental Health Before Prescribing These Medications

Editorial credit: marekuliasz / Shutterstock.
“These medications have become some of the most commonly used. The adverse events reported in this study serve as a reminder to physicians to carefully assess a patient’s mental health before prescribing these medications.”
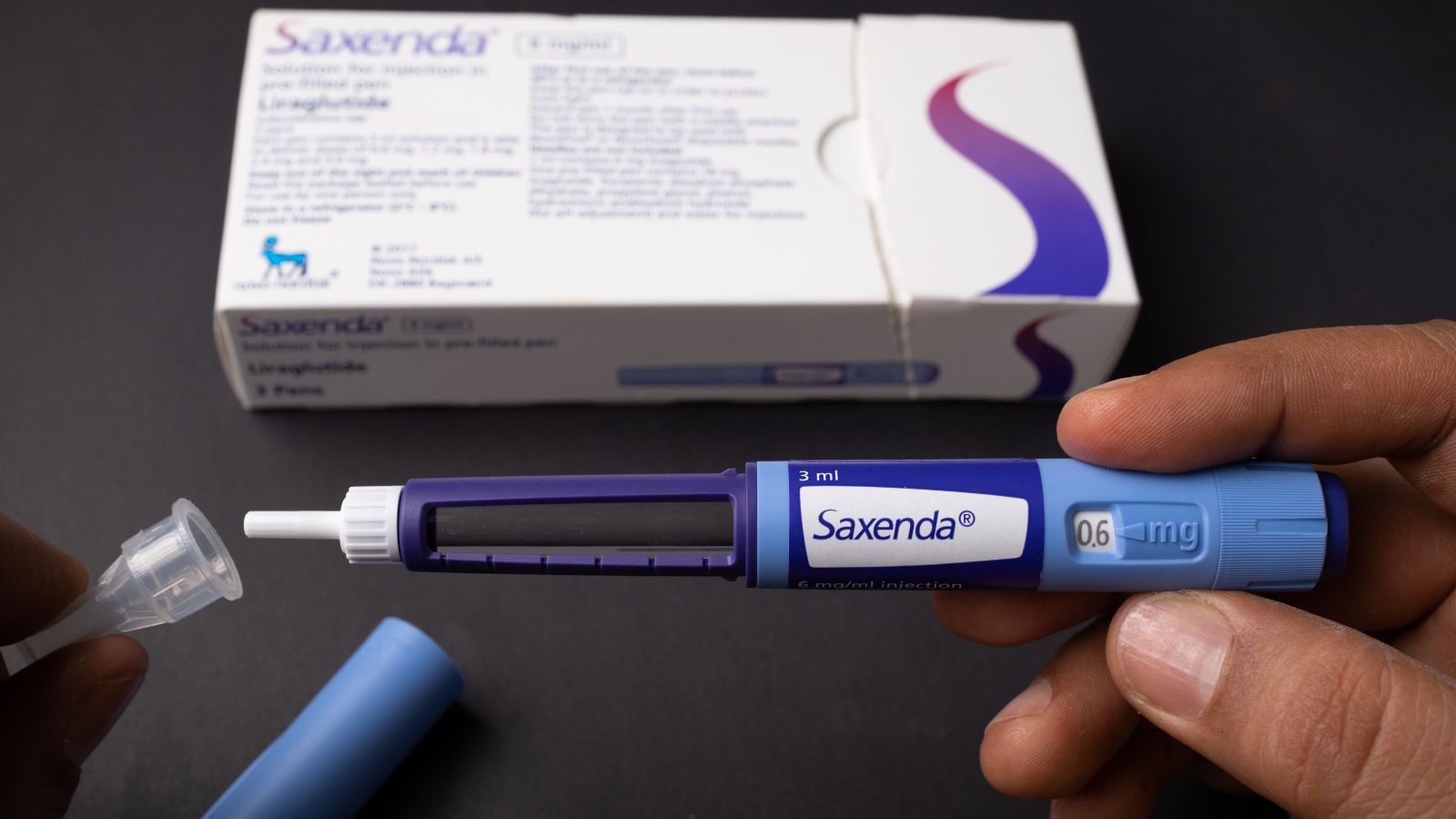
Case Reports Thoroughly Analyzed
Editorial credit: Mohammed_Al_Ali / Shutterstock.
Tobaiqy and co-author Hajer Elkout searched through EudraVigilance, the European Medicines Agency’s system for tracking and reporting drug adverse events, to arrive at these conclusions. Between January 2021 and May 2023, the two examined case reports about semaglutide (Ozempic and Wegovy) and the associated GLP-1 mimic medications liraglutide (brand name Saxenda) and tirzepatide (brand name Mounjaro).
A Serious Matter That Warrants Attention

Editorial credit: Jojje / Shutterstock.
There were 31,444 adverse events totaling 481 reports of adverse mental episodes were documented over this time. “According to the study’s findings, women made up 65 percent (n = 242) of the reports, while men accounted for 29 percent ( n=108),” Tobaiqy stated. “Fatal outcomes were predominantly among men (eight out of nine), resulting from completed suicide attempts and depression. This is a serious matter that warrants attention.”
Patients Should Engage in a Dialogue With Their Doctors

Editorial credit: Chinnapong / Shutterstock.
Tobaiqy stated that although further study is required to validate these findings, medical practitioners should give careful thought to their findings when deciding whether or not to provide these drugs to their patients. “Doctors should take into consideration any past suicidal thoughts or attempts by the patients,” he stated. “Logically, if patients have these mental health issues, they should engage in a dialogue with their doctors to discuss alternative medications or interventions. Patients are also encouraged to report any changes in mood, behavior, or negative thoughts to their doctors…and health authorities.”
We Must Take the Potential Harm Seriously
Editorial credit: mi_viri / Shutterstock.
“While I believe that the benefits of these medications, such as reducing the risk of cardiovascular disease, outweigh the risks, we must take the potential harm seriously, given the types and severity of the events reported.” This was also expressed by Michael Bloomfield, an excellence fellow, consultant psychiatrist at University College London, chairman of the Translational Psychiatry Research Group, and non-participant in the study.
Further Research Into This Is Needed

Editorial credit: metamorworks / Shutterstock.
“Given the potential severity of these side effects, I agree with the authors that further research into this is needed,” he stated to Newsweek. “It is difficult at this stage [to know] who may be particularly vulnerable nor how people can protect themselves without further research. In the meantime, people experiencing symptoms of depression including lowering of mood and suicidal thoughts during treatment with these drugs should consult their doctor. It may be that people with pre-existing depression or suicidal thoughts are more vulnerable to these potential side-effects, however that remains an open question.”
We Must Exercise Caution When Interpreting These Findings

Editorial Credit: fizkes / Shutterstock.
Furthermore, Bloomfield and Tobaiqy stressed that although these mental side effects were perhaps substantial, their frequency was quite low. “It is [also] unclear whether these patients had pre-existing mental health conditions when they started using these medications,” Tobaiqy stated. “[Therefore] we must exercise caution when interpreting these findings.” Professor of metabolic medicine at the University of Glasgow in Scotland, Naveed Sattar, stated that it is hard to determine if these mental side effects are a result of the drug or just a coincidence in the absence of definitive comparisons.
More Similar Studies Would Be Helpful

Editorial credit: ldutko / Shutterstock.
“There is no comparator of psychiatric risks in people with similar characteristics who had started on non-[GLP-1] therapy,” he stated to Newsweek. “Only by having some form of comparator does one get closer to the truth as psychiatric conditions are not uncommon in general. This does not mean further robust studies in this area are not needed. They are, but they should be high scientific quality and enable robust comparisons.” Sattar also cited a recent, extensive study conducted by researchers at Ohio’s Case Western Reserve University of Medicine that employed this comparison and discovered no increased incidence of suicidal thoughts following the usage of semaglutide-based medications. “More similar studies would be helpful,” he stated.
Not Aware of Any Reliable Evidence

Editorial credit: tadamichi / Shutterstock.
Newsweek got the opinion of Novo Nordisk, the maker of Ozempic and Wegovy, a pharmaceutical company, on Tobaiqy and Elkout’s most recent research. A Novo Nordisk representative informed Newsweek in a statement, “Analyses of spontaneously-reported adverse event data (such as that performed by Tobaiqy and Elkout) are informative but suffer from numerous inherent limitations which preclude drawing conclusions about association or causation […]. Novo Nordisk is not aware of any reliable evidence linking semaglutide with suicidal ideation or behavior, or with other adverse psychiatric events.”
FDA States No Evidence

Editorial credit: JHVEPhoto / Shutterstock.
“The FDA recently stated that their ‘preliminary evaluation has not found evidence that use of these medicines causes suicidal thoughts or actions.’ Similarly, our reviews of the clinical trials, including large outcome studies and observational studies, did not find an association between use of GLP-1 receptor agonist medicines and the occurrence of suicidal thoughts or actions. Novo Nordisk is continuously performing surveillance of the data from ongoing clinical trials and real-world use of our products and collaborates closely with authorities to ensure patient safety and adequate information to healthcare professionals.”
A Worldwide Misuse
Editorial credit: Sorapop Udomsri / Shutterstock.
“Novo Nordisk stands behind the safety and efficacy of semaglutide and of all our other GLP-1 receptor agonist medicines when they are used as indicated and when they are taken under the care of a licensed healthcare professional.” Tobaiqy did emphasize, though, that their research speaks to a larger problem with the usage of these medications for “off label” uses. “I believe there is currently a worldwide misuse of these medications by individuals seeking to achieve slimness, even when they don’t necessarily need them,” Tobaiqy stated. “Some counterfeit medications, available on the internet or black markets, may even lack active ingredients. Some are being sold without prescriptions, which poses a significant risk to patients’ health.”
These Medications Are Beneficial and Efficient in Treating Type 2 Diabetes and Obesity
Editorial credit: Proxima Studio / Shutterstock.
“My message is this: these medications are beneficial and efficient in treating type 2 diabetes and obesity. However, they should only be used under the supervision of medical professionals, whether they are endocrinologists or family doctors. All potential side effects, including mental health issues, must be closely monitored due to the novelty of these medicines.”
18 Things You Should Probably Stop Doing After Age 50

Image Credit: Shutterstock.
18 Things You Should Probably Stop Doing After Age 50
19 Products Marketed Almost Exclusively To Stupid People

Image Credit: Shutterstock.
19 Products Marketed Almost Exclusively To Stupid People
No Boomers Allowed: 15 States Where Retirees Are Not Welcome

Image Credit: Shutterstock.
No Boomers Allowed: 15 States Where Retirees Are Not Welcome
18 Disturbing Conspiracy Theories You Laughed Off But Were Actually True

Image Credit: Shutterstock.
18 Disturbing Conspiracy Theories You Laughed Off But Were Actually True
18 Everyday Phrases Unintentionally Reflecting White Privilege

Image Credit:Shutterstock.
18 Everyday Phrases Unintentionally Reflecting White Privilege